Additionally, protein epitopes simply afford more epitopes for antibody recognition which may result in more robust signal with a pAb, or greater potential for positive mAb clones. mAbs should be qualified via Western Blot during the cloning process to appropriately screen for application appropriate positive clones. However, using a protein antigen is not sufficient to guarantee a Western Blot functional mAb. Peptide antigens both increase risk of failing to provide enough non-conformational dependent epitopes and may not recapitulate the structure in its reduced form. To insure against this, both polyclonal and monoclonal antibodies intended for Western Blots should be raised against protein antigens. As reducing conditions denature proteins, some epitopes maybe lost due to conformational change as the protein is linearized. 
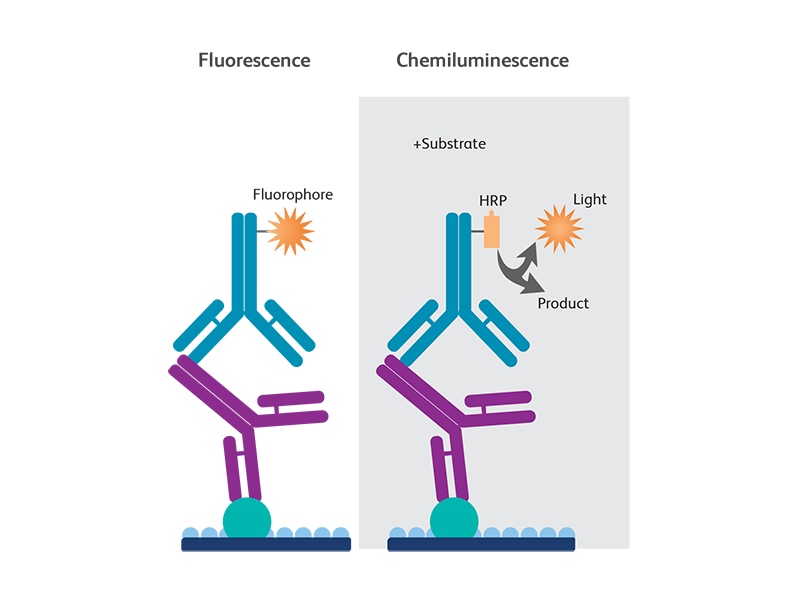
This is critical when performing a native, or non-reducing Western Blot, but serves reducing Western Blots as well. Peptide Antigen Using Antibodies Raised Against Protein Antigen vs Peptide Antigen in Western BlotsĪntibodies raised against protein, rather than peptide antigens, present the immune system with conformationally relevant epitopes. What can go wrong with antibodies when performing a Western Blot? Rock or agitate blots during incubation, insure full membrane coverage.Non-uniform spotting, irregular detection across membrane Antigen or protein A affinity purify antibodyĪntibody binding to blocking/binding buffer precipitate.Pre-adsorb against blocking protein/interfering protein.Pretreat gel or membrane with methanol or another solvent especially with polyvinylidene difluoride (PVDF)īlocking agent interference or cross reactivity (confirmed by secondary only control).Inefficient membrane transfer (confirmed with Ponceau S) Decrease blocking concentration and/or switch blocking agent (BSA, serum, non-fat milk, etc.).Concentrate sample to load at least 20 µg per wellĪntibody or antigen interaction with blocking buffer.While this technical note will speak to the aspects of antibody troubleshooting, there are many features to a Western Blot which can be modified and optimized as outlined in the chart below. What are some typical western blot problems? Antibodies may be directly detected with a signaling molecule (chromogenic, chemiluminescent, or fluorescent), or indirectly detected with a labeled secondary antibody. 
Immunoglobulins (IgG), usually IgG antibodies are used to specifically detect the protein(s) of interest, hence Western Blots also being referred to as "immunoblots". Moving the resolved proteins sample to nitrocellulose provides a durable substrate which may then be probed. Once the proteins in the sample have been sufficiently separated by their molecular weight, they are then ready for transfer. Larger proteins are retarded and remain closer to the loading wells at the top of the gel while smaller proteins travel further as the migration front, potentially off the gel if proteins are small and run long enough. Higher polyacrylamide percentage yields decreased pore sizes while increasing cross-linker percentage yields larger pore sizes. As they travel, proteins are restricted by pores whose size is determined by the polyacrylamide and cross-linker percentage of the gel. These negatively charged, linearized proteins then migrate away from the cathode, the negatively charged area near the loading region of the gel, and towards the anode. SDS both denatures folded proteins, allowing them to travel more easily through the gel, as well as impart a negative charge to them. Specifically in a Western Blot, samples of interest are loaded into a well of polyacrylamide gel (PAG) containing sodium dodecyl sulfate (SDS), to which an electric field (E) is applied, hence "SDS-PAGE".
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
